Annual Report 2017
Foreword
In 2018, the board will be celebrating its 25th anniversary. Its founding was motivated with the enactment of the Dutch Pesticides Act in 1962 and the establishment of the Dutch Pesticides Agency. In 1993, this agency officially became an advisory body and in 2000 it became an independent executive body. The legislative amendment in 2007 also included biocides authorisations and, as of that moment, the agency was called the Board for the Authorization of Plant Protection Products and Biocides (Ctgb).
At the time, a conscious choice was made to establish the Ctgb as an independent authorisations authority and independent executive body within the framework Act. The Ctgb has a public function with public authority and its own corporate capacity. The main aspect of our work is an independent evaluation based on factual and scientific evidence that takes place within the established Dutch and European frameworks. Alongside being necessary for the identifying functions we perform that are relevant for the departments and politics, this also safeguards a good relationship with the departments involved. The Ctgb is cost-effective, as we only charge applicants the costs that we have incurred for the evaluation process. This makes the Ctgb one of the few authorisation authorities that can adjust its capacity to the development in demand in Europe.
When reflecting on our history, we can identify four trends: the transition from a national focus to a European focus, the transition from chemical to more integrated and green, from broadly effective to more targeted, individualised effectiveness (digital) and the increase in public interest.
For biocides and plant protection products, the trend towards European harmonisation can be identified in various fields. The number of substance and product authorisations under the Biocidal Products Regulation (BPR) increased significantly, and the first Union authorisations of biocidal product families evaluated by the Ctgb were confirmed in the Biocidal Products Committee. We noticed more reservation in the new applications for plant protection products and substances.
Within Europe, we work closely with the European Commission (EC), the European Food Safety Authority (EFSA) and the European Chemicals Agency (ECHA). We have agreed to run pilot projects for the disjuncture between biocides and plant protection products in order to achieve an accountable substance evaluation in a simultaneous, coherent and, perhaps in the future, joint evaluation process that will extend to the evaluation and authorisation of such products. This aligns well with the need and tendency to blur the boundaries between biocidal products and plant protection products where possible and, indeed, where safe. Moreover, this also reflects the wish of both fields to be open to new developments such as plasma and cold plasma applications.
In a pilot project run together with the EFSA, we will investigate whether it is possible to agree on a package of measures during the phase of substance evaluations and authorisations. For example, this could prove beneficial for the greenhouse horticulture sector and areas with mountains or river deltas, so that national authorisation authorities can use these for the authorisation. This can significantly reduce the evaluation periods for products and contributes to harmonisation. A few nationally specific elements will continue to play a role, but these will be fewer. Part of the work that currently falls in the phase of evaluation and authorisation of substances will then fall under the substance approval phase.
On the initiative of several directors and with the permission of the EC and the EFSA, the opinion of the EFSA will be formulated at the moment that an evaluating Member State circulates the drafts of the risk assessment in Europe. This will mean that all commentary will be collected in one round. This will be decided shortly and will lead to a more efficient and shorter process.
The second trend of becoming more sustainable and the transition from chemical products to more integrated and green products is especially noticeable for plant protection products. During the evaluation process we make a contribution by providing guidances and manuals for green products, low-risk products and biostimulants. The European criteria for low-risk products have now been established. We have committed ourselves to achieving a harmonious working method for the evaluation of the efficacy of green products. There is also a list of European basic substances and, together with the Green Deal and the current pilot project for system management, you can observe a steady increase in the possibilities for authorised products and substances on this side of the spectrum. However, we’re not there yet. In the coming period, the Ctgb will be asked to further develop this process and to apply it to the evaluations.
The third trend is the transition from broadly effective to more targeted, individualised effectiveness. We are investigating ways to correct outbreaks in the context of integrated pest management, via the use of “green” products, or even with spot-wise use of conventional products, where necessary. There are currently several active pilot projects to investigate how this can be monitored and measured using a type of environmental yardstick . Using this information and a responsible evaluation and authorisation process, we can then allow the use of a specific product under well-defined circumstances as a spot-wise correction product. With the current development in ICT and digitisation, it must be possible during the authorisation to accurately determine the measures required for responsible use. The specification of techniques for use and digital farming are developing rapidly towards individual plants or crops as a basis for a decision for the use of certain products. In the future, plant health will be just as specified and individualised as pharmaceuticals and medicine. The objective is to significantly reduce the amount of chemical products used. In the end, this is an effect that may well have much more impact and may yield more results than the ‘for or against chemicals’ debate.
Here, the fourth trend of increasing public interest is of relevance. This is becoming more apparent in the number of objections and appeals. Public access and maximum transparency is one of our basic principles and only business information and confidential information is protected. The specification of this is currently awaiting the court decision. As the Ctgb, we also believe in clear rules. In the last few years we have improved the way in which we present ourselves to external parties, the way in which we explain our decision-making process and with whom we are in discussion and are in contact with. The authorisation database and website have been updated accordingly. We make ourselves accountable to the public in a general sense through our website and the annual report. This is also observed and appreciated by our field.
The main aspect of our work is risk assessment. However, in recent years, the tendency to make assessments from a hazard-based perspective has increased. This is a cause for concern, as we base our evaluations on facts and science through which the risk evaluation and its associated standard will lead to responsible use for humans, animals and the environment. This is the very foundation of our work. Exposure and potential danger together determine the actual risk. The Ctgb does not only authorise, but it also denies applications. Approximately 15% of applications for authorisation and use are denied every year. So we are not just there to hand out authorisations. But when considered, the part of society that does just take into account the hazard and not the risk does keep us sharp and critical.
This all demands a great deal from the organisation, which is further burdened by the effects of Brexit, a stagnation in current applications for plant protection products and the large increase in biocidal product applications where the moment of submission is not always as predictable. This requires great flexibility of the staff and the organisation to do the work as coherently and quickly as possible.

This was made a little more complicated, rather than easier, in the second half of last year, despite being better equipped through the new Grip on Planning system. However, we ended last year with a tentative positive result that we are quite happy about and was achieved thanks the commitment of all our staff.
The board bid farewell last year to Arnold van der Wielen who reached the end of his last term as a deputy board member. For many years he served the board with his extensive knowledge of chemicals and substances for biocides.
Johan F. de Leeuw, Chair
Annual report: a summary of 2017
The Board for the Authorisation of Plant Protection Products and Biocides (Ctgb) is the Dutch authorisation authority for plant protection products and biocides. Its most essential task is to evaluate the safety and effectiveness of products for the agriculture and horticulture sectors and products such as disinfectants for hospitals and home-use, antifouling paints or products that combat pests and to make a decision whether to authorise them or not. The Ctgb authorises a product only if it is effective when used safely and is not harmful for people, animals and the environment.
In 2017, the Ctgb made 184 decisions for applications for plant protection products and 65 for biocides. These decisions concern multiple uses of a product. The Ctgb authorised 83% of the requested uses for plant protection products and 84% of biocidal product applications for use. In comparison, 17% of plant protection product and 16% of biocidal product requests for use were rejected or were withdrawn by the applicant due to the risks identified by the Ctgb. For 59% of plant protection products and 33% of biocidal products further instructions were defined prior to authorisation, so that consumers and growers can trust that the products that they purchase and use are safe when used according to the instructions. The number of authorised, rejected and amended applications did not differ significantly to previous years.
The organisation became more stable. Staff became more organised, which created space to maintain and develop knowledge. For the first time in years the available budget for knowledge development was fully utilised. Large projects were completed, including the Middelen-, Stoffen- en Toepassingendatabank (products, substances and uses database), which is a database with information about uses and the Grip op Planning planning system with which the Ctgb hopes to better inform applicants and more accurately predict the processing times.
It appears to be more difficult to get a grip on external factors. The progress of the risk assessment for the applications under the BPR initially followed the planning, but later in the year it became clear that the European coordination process for these applications took more time than expected. Uncertainty about the effects of Brexit on the demand for the evaluation capacity of the Ctgb also grew. The number of biocides applications in particular increased. This was less clear in the applications for plant protection products, as the increase in the demand was veiled by the reduction of the number of applications on a European scale. Applicants spent a lot of time and energy in renewing their substance applications, as nearly all application dossiers had to undergo a complete re-evaluation. A number of substances were no longer authorised, which meant that the products that contain these substances also lapsed. The number of applications for new substances also decreased, also for green substances. Attention was especially focused on maintaining the current range of substances.
Harmonisation
The Ctgb performs its work within the European frameworks for the regulation for plant protection products and the regulation for biocidal products and adheres to national legislation where applicable. It works together with the European Food Safety Authority (EFSA), the European Chemicals Agency (ECHA), the authorisation authorities in other Member States and with the various ministries in the Netherlands, the Netherlands Food and Consumer Product Safety Authority (NVWA), the Human Environment and Transport Inspectorate and research institutes such as the National Institute for Public Health and the Environment (RIVM). Harmonisation is an important objective within European collaboration. That means that products are evaluated and authorised in the same way in all Member States of the European Union, so that producers, farmers, horticulturalists and other users have the same products at their disposal and can compete fairly.
The European regulations have not been worked out in detail and guidance documents are continuously being developed for further specification. The Ctgb actively contributes to this development. In the past year, the Ctgb was focused on harmonising the evaluation of antifouling paints for recreational boating. While the deadline for the paint industry to submit application dossiers was approaching, there was no European guidance available. Prompted by the Ctgb, the Health and Safety Executive (HSE) in Great Britain and the RIVM working together with a number of authorisation authorities, developed calculation sheets for both saltwater and freshwater marinas. In autumn, the ECHA published the guidance document and the calculation sheets so that applicants could make calculations for antifouling products on yachts, sail boats and motorboats.

Developing EU policy
New EU policy also requires further formulation by the Ctgb. After the European Commission indicated that it regards disinfectants to protect crops as plant protection products, the Ctgb further developed its policy for the authorisation of biocides that can be used for this purpose. After re-registration, new biocides and existing biocides may only be used in agriculture if they are authorised for general hygiene. A biocide specifically intended for the protection of plants or plant products, for example for the disinfection of greenhouses, is therefore no longer possible. For this type of use an authorisation as plant protection product is required.
In the summer, the Ctgb established a policy for biocides and plant protection products containing pheromones or attractants and clarified when they do and do not need an authorisation. This policy is compatible with the European guidances and agreements. Semiochemicals are chemicals that release substances that attract, repel or disrupt the behaviour of plants, animals or other organisms. Various biocidal products and plant protection products contain these chemical substances. Depending on their purpose, they may require authorisation to be used as a plant protection product or biocidal product. Traps containing attractants do not need to be authorised under the EU plant protection regulation as long as they do not contain any other chemically active substance, as attractants are not considered to be an active substance. The BPR does not require authorisation for traps intended exclusively for counting pests and insects for monitoring purposes, but does require authorisation as soon as these traps are used for pest control.
Hand and skin disinfectants (product type 1, human hygiene) were authorised under the previous Dutch testing framework as 'agents to combat bacteria, yeasts, moulds and viruses'. This is known as a full virus claim. In practice, it was assumed that these products were active against all known viruses. Under the new European assessment framework, three degrees of efficacy will be distinguished from May onwards: full virus claim, limited virus claim and activity against enveloped viruses only. In terms of efficacy, the limited claim lies between the full virus claim and the claim of being active against enveloped viruses only. From now on, the products for hygienic hand disinfection must be effective in 30 to 120 seconds. For surgical hand disinfection, contact times of up to five minutes are permitted.
In January, the ECHA published a guidance document for disinfection by-products (DBPs), most of which was developed in the Netherlands by RIVM, which comes into effect on 1 January 2019. DBPs are formed mainly from the use of halogenated biocides and can pose a risk to human health and the environment. The guidance on DBPs applies to three product types: disinfection of swimming pool water and wastewater (PT2), cooling water (PT11) and paper production (PT12). This means that applicants have to submit a risk assessment for these product types. The human health risk assessment is based on a set of known marker DBPs, using consensus health-based limit values and DBP concentrations. The guidance is focused on disinfecting swimming pool water (PT2), because human exposure to DBPs is most relevant here. The environmental risk assessment is predominantly focused on the determination of the risks of DBPs directly or indirectly present in surface water. This also applies to wastewater and paper production.

RUB list
The withdrawal of the Pesticides Exemption Scheme (Regeling Uitzondering Bestrijdingsmiddelen, RUB) authorisations in the Netherlands is an exception. All plant protection products and biocides must be evaluated according to European directives and regulations. However, the Netherlands still had a few exceptions to this. These were substances with uses that were authorised based on the old Pesticides Exemption Scheme In 2012, it was agreed that the RUB products would either be authorised on the basis of the European regulation or would be withdrawn from the market. Some of these products have held an exceptional position in the Netherlands for many years and the imminent withdrawal of the regulation caused considerable concern among users. The Ctgb therefore made every effort to inform and guide applicants in the submission of a regular application. It presented the draft plan to bring the RUB products under European legislation for consultation and organised an information session. At the end of October the board agreed to the definitive process. Non-registered RUB products will be withdrawn from the market after 1 October 2018 and may no longer be used after 1 October 2019.
Union authorisation
Union authorisations for biocidal product families are a novelty. The BPR enables the single authorisation of products or product families throughout the whole of the European Union. These are the Union authorisations granted by the European Commission. In June 2016, more than half of the applications for these new Union authorisations (27 of 44) were submitted to the Ctgb. The Ctgb thus took the lead in Europe: at the end of 2017, the Biocidal Products Committee in Helsinki discussed the first two Union authorisations which will be adopted in the first half of 2018.

Secretariat of the Central zone
For plant protection products, eleven of the thirteen Member States attend the meetings of the Central Zone Steering Committee and the Directors Council Group. One of the Member States shall hold the rotating chairmanship of these consultation structures for a period of one year. In the Directors Council Group, the idea for a Secretariat of the Central zone, staffed by two Member States, was formulated in order to reduce the administrative burden. Germany and the Netherlands offered to participate in the pilot starting on 1 January 2017. Due to the good results, the pilot was extended by another year in September. Germany and the Netherlands continue to serve on the Secretariat. As the consultative processes now run smoother, the Member States hope that harmonisation will be easier to achieve.
Sustainability
The Ctgb responds to the demand from the government, society and the agriculture and horticulture sectors for more sustainable products, integrated pest management (IPM) and low-risk products. It was one of the partners of the Green Deal for plant protection products which was concluded in mid-March in the Ctgb office with the presentation of the final report to the press and the public. The aim of this Green Deal was to carry out nine concrete evaluations of green plant protection products – two substances and seven products – to see how that process could be accelerated or simplified. Six products were authorised in the period of this Green Deal. The Ctgb set up a special GreenTEAM and appointed a coordinator for organic products. The authorisation procedures appeared to be faster and significantly cheaper, as green substances are often less complex. The applicants are also encouraged to apply for substances for as many crops and infections as possible.
In July, the Ctgb formulated the special Evaluation Manual for green products (biopesticides) to help applicants in submitting an application dossier. High-quality application dossiers are easier to evaluate by the Ctgb. The manual is an addition to the existing guidance documents. The Evaluation Manual Biopesticides describes how the Ctgb interprets and applies the guidances and data requirements in the risk assessment of active substances and in the evaluation of applications for substances based on microorganisms, plant extracts and semiochemicals. The new manual is a tool for evaluating green substances consistently and can make the process much clearer for applicants. Due to a great deal of interest, the Ctgb organised four workshops to explain the manual in September and October.

Low-risk substances
In September, the European Commission set new criteria for low-risk substances to encourage the use of these substances in plant protection products. The criteria are now better suited to microorganisms, for example. New low-risk substances will be authorised for a longer period of time than other active substances. This period will be fifteen years instead of ten. For dossiers for zonal applications for products based on these substances the statutory period for evaluation is 120 days with an extension of a maximum of six months for the applicant to answer additional questions. This only applies if all active substances and all uses of the product are low-risk. In the European context, it is now being determined which procedure can be used by the competent authorities to comply with the 120-day assessment period for zonal applications. In Europe, the Netherlands took the lead in establishing harmonised and realistic requirements for the efficacy of green products. Many of the green products meet the criteria for low-risk products.The basic substances are a separate category of low-risk substances. These may not be sold as plant protection products but are able to protect crops. Beer, which was included on the RUB list in the Netherlands, and ground mustard seed were added to the European list of basic substances in October. This list now contains more than 20 substances that may be used for plant protection under specific conditions. It is generally assumed that using these basic substances entails little risk and they may not affect the endocrine system, the nervous system or the immune system.

Authorisation as a basic substance may be an alternative for some products that previously fell under the revoked RUB. Basic substances are authorised at the European level. Each interested party can submit an application, including the Member States themselves. For example, beer has been applied for by a French research institute for organic agriculture (ITAB), which has previously applied for various European authorisations for basic substances. The European Commission approves a basic substance for certain uses following scientific evaluation by the European Food Safety Authority (EFSA) and consultation of all Member States. This procedure takes approximately one year and the authorisation of a basic substance has no expiration date.
Safety
If, despite the regulations, unacceptable risks for humans, animals or the environment are found to occur, the Ctgb intervenes. Despite regulations and measures, too high concentrations of imidacloprid (a neonicotinoid) were still found in the water in the greenhouse areas. The Ctgb banned the use of imidacloprid-containing substances in greenhouses as of 15 March, unless a grower can demonstrate that they purify at least 99.5% of the active substance from the wastewater. This must be verifiable and guaranteed with a certificate. The extension of this measure was not possible due to the risks for water organisms.
The necessary purification technology was available and the certification could be provided by existing quality marks. For purification techniques, the Ctgb sought to adhere to the Hoofdlijnenakkoord waterzuivering in de glastuinbouw (general agreement for water purification in greenhouse horticulture). The purification of wastewater fits in with the future objective of achieving zero-emissions greenhouse horticulture. With just one installation, growers can meet the purification criteria of the general agreement and, if registered with the Ctgb, of the additional purification requirement of 99.5% for imidacloprid. A pilot project was launched for growers with closed greenhouses who do not discharge wastewater to see how this system could be secured in such a way to ensure the responsible use of imidacloprid by these growers. Moreover, in order to prevent abuse, the use of all imidacloprid-containing products intended for use in greenhouses were included under the system of controlled distribution. Later in the year, the Beoordelingscommissie Zuiveringsinstallaties Glastuinbouw (assessment committee for purification plants in greenhouse horticulture) started to determine the purification efficiency, as a result of which horticulturists knew which installation they could buy and the regulatory authority knew whether a horticulturist achieved the mandatory minimum purification efficiency.
In addition to the discussion on neonicotinoids, the public debate in 2017 was dominated by the imminent renewal of glyphosate and the fipronil issue. The Ctgb, once again, made a positive recommendation for glyphosate to the minister regarding the vote in the Standing Committee on Plants, Animals, Food and Feed (SCoPAFF). It also concentrated more effort on explaining its working method and policy to external parties. The deputy director gave several radio interviews and was interviewed for the science supplement of a national newspaper. The director regularly wrote columns for an agricultural magazine and the Ctgb E-zine (e-mail newsletter), which were received well and picked up by the trade journals. At the end of the year, an interview with the chair of the board and the director was published in an agricultural magazine.
Following the fipronil issue about red mite control at poultry farms that dominated the news for weeks, the Ctgb received many questions about green products that control red mite. The Ctgb therefore published several notices on its website in which it explained how you can see from the number on the label whether a substance has been authorised and that fipronil is authorised for the treatment of certain seeds and as a gel against cockroaches, but not as a substance against red mite to be sprayed in the sheds.

Appeals, objections and requests under the Government Information (Public Access) Act
The number of objections and requests received for interim measures increased and the number of appeals did not really decrease. There was also persistent legal complexity: the Trade and Industry Appeals Tribunal again announced that it would be asking preliminary questions in a case concerning parallel licences. A new development was that extensive requests under the Government Information (Public Access) Act (Wet openbaarheid van bestuur, Wob) were submitted by journalists, and also to the ministries and the RIVM and sometimes on subjects on which work was jointly carried out, such as endocrine disruptors. (Also refer to Chapter 4, p. 31.)
The court's handling of the extensive appeals and objections in the neonicotinoid cases and the requests for disclosure contained therein has been slow. A pretrial hearing was held by the Trade and Industry Appeals Tribunal in October. A substantive hearing was also convened for the beginning of 2018 about the requests for disclosure that led to preliminary questions and the November 2016 ruling of the European Court of Justice. However, a final settlement of these cases is not expected in the short term. These cases continue to put a considerable strain on the deployment of people and resources, including those of the parties involved.
The Ctgb is in contact with many parties in order to be properly informed about all facts and interests that play a role in authorisations. In order to be able to take decisions with due care and independence, it is necessary to have a good understanding of these matters. The Ctgb has these contacts not only with the admission holders, but also with industry associations, users and NGOs. The agendas for the consultations with these parties are now available on the website in order to make transparent which parties we are in contact with.
Innovation
Innovation was the theme of the annual client day held on Thursday 15 June, which was visited by more than 200 participants. The lectures, presentations and discussions on plant protection focused on habitat management as support for sustainable plant protection, the use of new resources based on RNAi technology and the possibilities and impossibilities of natural plant protection in combination with chemical substances. During the biocide sessions, cold plasma was discussed as a disinfectant and anti-pathogen agent and UV light as an option for disinfection but also to prevent the growth of marine plants and animals on the hulls of ships. In both biocides and plant protection, attention was paid to the growing importance of an integrated approach that addresses not only the pest, but also its potential for emergence and sometimes its actual breeding ground. This principle can be applied in agriculture and horticulture as well as in combating nuisance caused by pests such as rats and mice.
New authorisations database and website
In recent years, a great deal of effort was put into updating the authorisations database and creating the new website. Both went live in September. The new authorisations database, which is an investment by the Ctgb, the Netherlands Food and Consumer Product Safety Authority (NVWA) and the Ministry of Economic Affairs, has been supplemented with information about applications. Now, visitors to the database can also find relevant authorised substances when searching for a specific application. This will make all public data, including the authorisation decision and the assessment report, easier and quicker to access. The technical data of the database have been made available to app developers who can develop their own apps on the basis of these data. The renewed database is thus in line with the government's policy of making raw data available for use by private individuals and the business community.
In addition to structured, specialist information for applicants, the renewed Ctgb website now also contains more easily accessible information on biocides and plant protection products for interested parties. This includes background information on current themes, such as working together on the sustainable and safe use of plant protection products and biocides. The database and website immediately received many visitors.
Groundwater Atlas
Wageningen Environmental Research (Alterra) and RIVM published a Groundwater Atlas for pesticides. This is a database containing measurement data. The Groundwater Atlas shows which plant protection products, biocides and their metabolites are found where and in which concentrations. The measurement data come from the observation wells of drinking water providers and provinces covering the period from 1990 to the present. Provinces and drinking water providers in the Netherlands regularly take samples of the groundwater to assess its quality.
The Ctgb wanted this information to be available and to be able to use it more explicitly in the assessment of the authorisation of plant protection products. For example, it can test the theory of assessing whether safe use is possible by comparing the predicted concentrations against the actual concentrations in the groundwater. To this end, the measurement data must be made public and must include information on aspects such as land use and the source of the groundwater. This makes it possible to detect whether a plant protection product, despite being used in accordance with the regulations, still exceeds the authorisation standard. In this way, the Ctgb, the provinces and drinking water providers are working together on the safe use of plant protection products and good groundwater quality.
Sharing knowledge
The Ctgb shared the knowledge gained in the risk assessments and development of guidances with European bodies, applicants, users, fellow authorisation authorities and interested parties in consultations, at conferences, conventions, via the website and in workshops and information meetings. In addition to regular workshops and information meetings, it also organised an information meeting last year on the RUB resolution and workshops on the Evaluation Manual Biopesticides.
In June, the Ctgb received a group of six scientific evaluators from the Kenyan Pest Control Products Board (PCPB) for five days. PCPB is working on a project in partnership with Wageningen Environmental Research (Alterra), and the Kenyan evaluators were especially interested in the assessment of low-risk products and microorganisms in particular. The GreenTEAM organised a customised workshop and took them on a tour through a company working with these products, after which they were better equipped for low-risk product applications in Kenya.
A Ctgb project leader for biocides, who had previously chaired the Coordination Group for two years, was seconded to ECHA for one year. And at the end of the year, the EFSA signed a cooperation agreement with the Ctgb. EFSA sought cooperation partners with knowledge of tasks within its mandate. This cooperation should facilitate the exchange of knowledge and experience between EFSA and the authorisation authorities of the different Member States and improve the efficiency of the European approval and authorisation process. The Ctgb mainly tendered for components in the field of greening. It was offered a four-year agreement.
Image and evaluation
According to the Framework Act, an independent administrative body such as the Ctgb must be evaluated periodically. This is also in keeping with the Ctgb's commitment to quality – the organisation is ISO-9001 certified. In 2016, the Galan Group evaluated the work of the Ctgb over the 2012-2015 period. The State Secretary for Economic Affairs sent the Ctgb Evaluation Report to the House of Representatives of the Netherlands in August 2017. In his letter he concluded, ‘...that the report of the Galan Group gives a positive picture of the development of the effectiveness and efficiency of the Ctgb in the period 2012 up to and including 2015.’ This was also reported in agricultural trade journals. The report also contained a response from the Ctgb to the House of Representatives of the Netherlands, in which it responded and indicated which actions had been taken in the meantime to further improve effectiveness and efficiency, and to improve finances and business operations.
Every four years, the Ctgb also commissions an image research study. At the end of 2016, applicants, consultants, NGOs and local authorities were asked about how they perceive the Ctgb. When asked for a spontaneous response, the respondents described the Ctgb as ‘expert, professional and ... official.’ The emphasis differs per subgroup. The overall trust in the Ctgb was more than satisfactory.
Organisation
The Ctgb is a professional organisation with happy and dedicated employees. In 2017 this resulted in the award of a best employer 2017-2018 quality mark from Effectory and Intermediair. The Ctgb has an open-door policy and people get along well and there is a great atmosphere within the organisation.
The new organisational policy came into effect on 1 January 2017. The organisational structure was further adjusted and the management team was structurally reduced from five to four members. The functions have been redesigned and adapted and research has been carried out into a more systematic structure for more internal mobility, flexibility and roles.
In 2017, the organisation grew steadily and in a controlled manner – especially within the primary departments – in order to be able to accommodate the larger number of applications, most notably for biocides. Despite the recovering economy, the Ctgb succeeded in filling all vacancies. Staff turnover was less than last year and proceeded in a more controlled manner: there was no unexpected outflow of staff. This growth naturally had an impact on the incumbent organisation. For example, the Ctgb invests intensively in training scientific assessors. This is a process that lasts at least a year. At the end of 2017, the staff consisted of 154 employees (138.8 FTE), which is six more than in 2016. Under the same circumstances and with limited growth expected in 2018, the staff is in order and the organisation will be able to achieve the objectives for the coming years.
The Grip op Planning planning system
In order to optimally control the primary process and improve the predictability of the progress of applications, the Ctgb started working according to a new planning concept in 2017. A corresponding planning application has been put into operation, and the operational planning for all departments is now proceeding according to this new working method. Work on the modules for tactical and strategic planning will continue in the first half of 2018. Once these have also been completed, project leaders and account managers will be able to better inform applicants and predict the lead times of applications.
Financial results
The Ctgb concluded 2017 with a positive operating result of €45,000. In the original budget, a positive operating result of €150,000 was taken into account. This was adjusted downwards in the course of the year when it became clear that not enough billable hours could be made to achieve this. The higher-than-expected positive outcome was due in part to an intense focus on the expenditure on operating expenses, the release of a number of resources from non-cost-covering applications that have been completed and the income from the annual reimbursements that exceeded expectations. The number of authorised products at the reference date of 1 February was higher than budgeted, as a result of which more income was generated. The positive financial result does not yet lead to a positive equity in 2017. The operating result for 2017 will be added to the general reserve, which will reduce the negative equity. The desired size of the general reserve is 10% of the average annual turnover over the past three years.
Risk management
For 2017, the Ctgb had formulated possible business risks that could influence the realisation of the work plan. The possible risks identified were 'insufficient staff capacity and excessive staff turnover', 'harmonisation bottlenecks' and 'consequences of Brexit’. These risks have been established and the consequences of the Brexit for the Ctgb can be assessed.
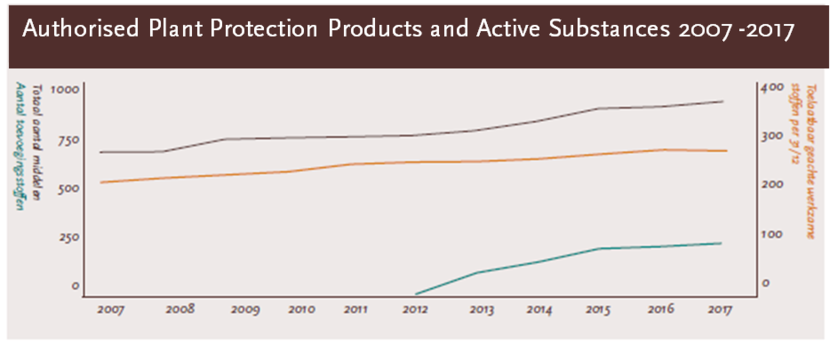
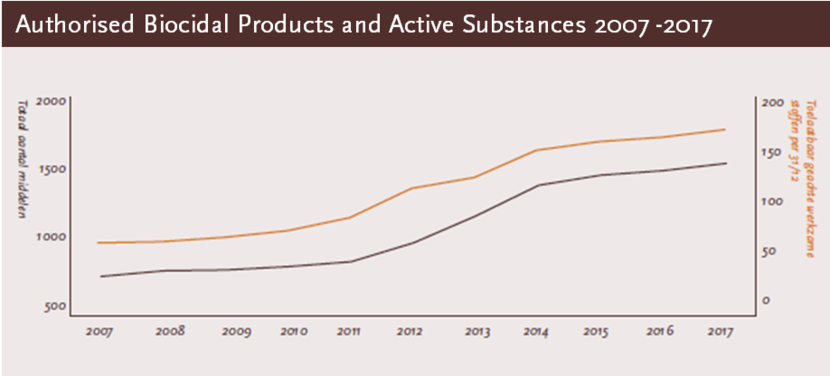
Authorised products and active substances